Iso 13485 Auditing
If you are searching about How to Implement ISO 13485:2016 and ISO 9001:2015 in One Management you've visit to the right web. We have 9 Images about How to Implement ISO 13485:2016 and ISO 9001:2015 in One Management like ISO 13485 and 21 CFR Part 820 Internal Audit and Gap Analysis Checklist; Planning an ISO 13485 QMS audit? Steps for preparing. and also How to Implement ISO 13485:2016 and ISO 9001:2015 in One Management. Here it is:
How To Implement ISO 13485:2016 And ISO 9001:2015 In One Management
 13485store.com
13485store.com qms
Manufacturing ISO 9001:2015 â€" Axeon Corporation
axeon
Top ISO 9001 Consultant In Jonesboro; Arkansas (AR)
 www.iso9001arkansas.com
www.iso9001arkansas.com jonesboro auditing cni
ISO 45001 Publication Delayed - Batalas
 www.batalas.co.uk
www.batalas.co.uk delayed stamp delays text delay fda deadline deeming vector vaping360 shutterstock
ISO 13485 And 21 CFR Part 820 Internal Audit And Gap Analysis Checklist
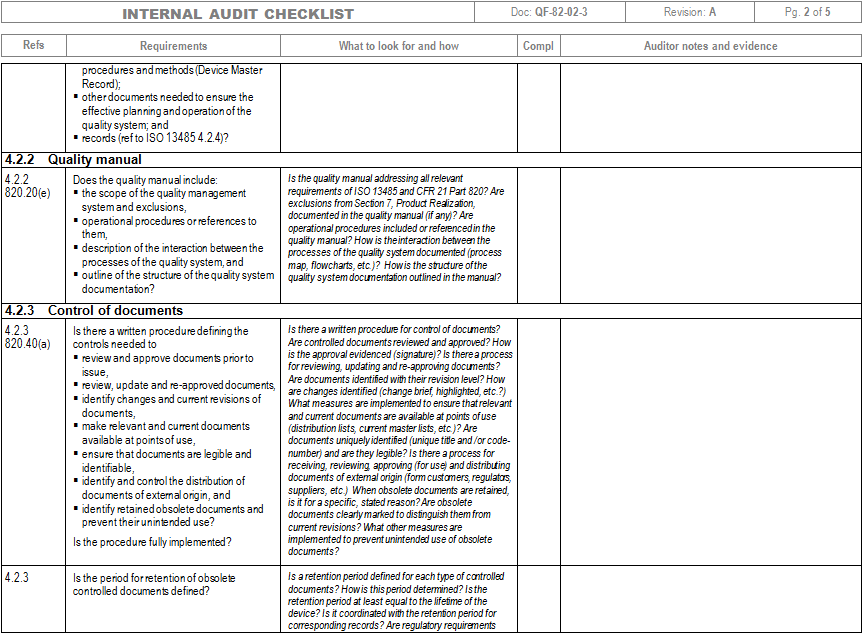 www.zenonhost.com
www.zenonhost.com audit checklist iso cfr internal analysis gap sample management document control fda file qsr
ISO Standards | URS Certification | A Listly List
 list.ly
list.ly certification
Planning An ISO 13485 QMS Audit? Steps For Preparing.
audit iso table qms detailed planning plan internal sample matrix auditing oriel team learn want
ISO 16949 | ISO/TS 16949 | TS16949
iso ts automotriz norma certification certified 2009 steel iatf requirements nevatia facilities wire stainless bengaluru surveillance audit service 2002 precision
Internal Audit Schedule Template
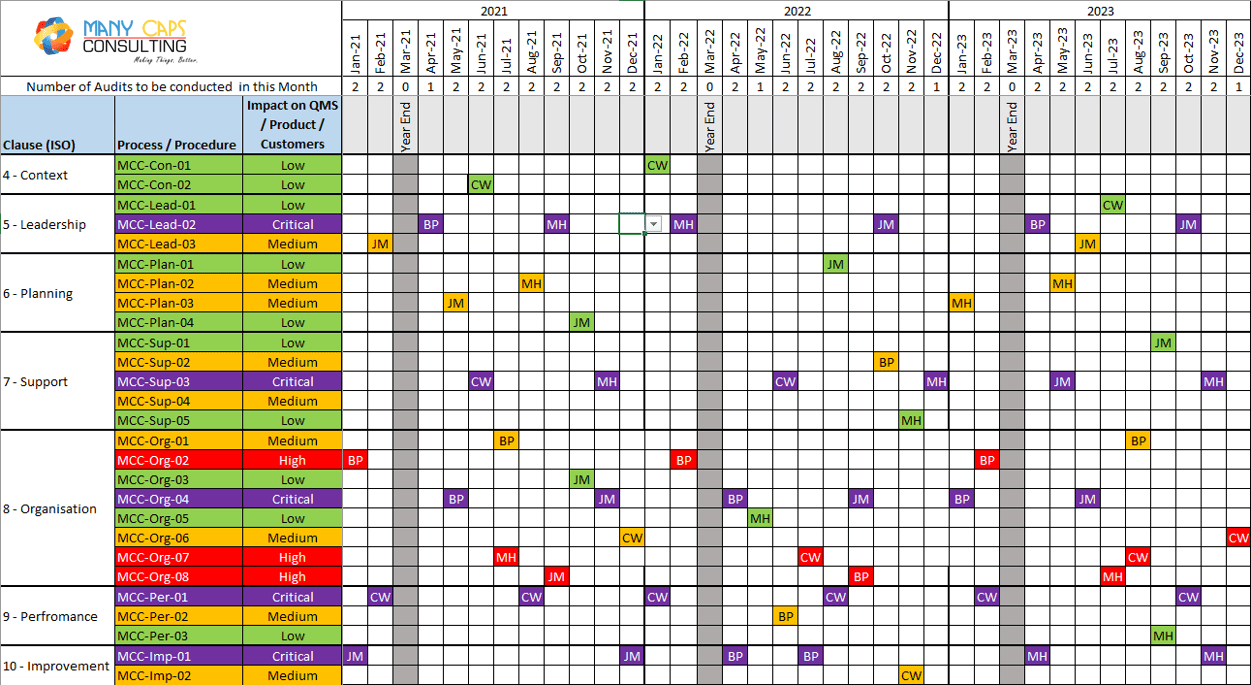 www.manycaps.com
www.manycaps.com audit internal
Audit iso table qms detailed planning plan internal sample matrix auditing oriel team learn want. Iso standards. Internal audit schedule template
Komentar
Posting Komentar